|
The greater the separation of charges between the atoms in bonds, the greater is the value of. 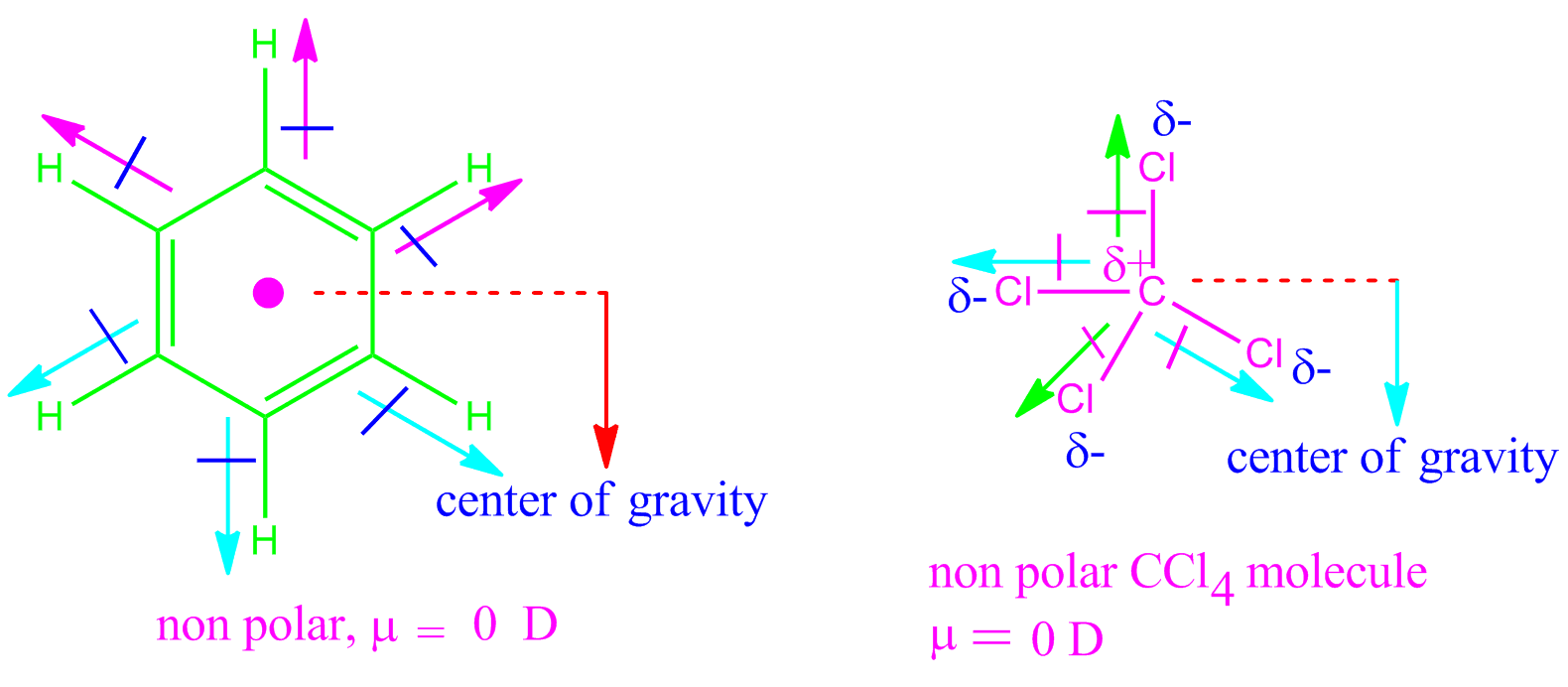 
where q is the amount of charge and d is the distance between the two charges. When placed between the charged plates, it will neither move toward the positive plate. Dipole moment arises in a polar bond be it ionic or covalent. The magnitude of the turning force is given by the formula. With no net force, the center of mass of the dipole will not accelerate, but there will clearly be a torque exerted on this object. trans-1,2-dichloroethene has no dipole moment and is nonpolar, while cis-1,2-dichloroethene has a dipole moment and is polar. Strategy: For each three-dimensional molecular geometry, predict whether the bond dipoles cancel. When you place a molecule with an electric dipole in an electric field, a force acts to turn the molecule so that the positive and negative ends line up with the field. In a ferroelectric material, there is a net permanent dipole moment, which comes from the vector sum of dipole moments in. Similarly, on the axis of the dipole (but outside it), the field points in the same direction as the dipole, again getting weaker the further one gets from the charges.Trending Questions What does the purity of a ring have to be to be real gold? What is the haemolytic action of saponin? What types of elements come together to form ionic compounds? What is the difference between acidic oxide and basic oxide? Boron has how many protons in its nucleus? How does sulfuric acid turn into acid rain? When we put some ice in a glass we see some water droplets on its outer surface. Ernest Zinck 9 years ago A moment is a measure of a turning force about an axis. Here there is an electronegativity difference between S and Cl where Cl is more polar and the dipole moments point towards Cl. Which molecule(s) has a net dipole moment H 2 S NHF 2 BF 3 Answer.  
This is because in BeF2 molecule, the bond. Notice that along the plane perpendicular to the axis of the dipole and midway between the charges, the direction of the electric field is opposite that of the dipole and gets weaker the further from the axis one goes. Explanation: Net dipole operates on the same idea - but it focuses on the direction and magnitude (vector) of the dipole moment. There exist two individual bond dipole moments, which cancel each other resulting in the net dipole moment zero.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
